Equine synovial fluid protein equalization via combinatorial peptide ligand libraries
Authors: Pablo Fueyo, Marco Galleguillos, Cristóbal Dörner, Pedro A. Smith, Francisca Godoy and Héctor Adarmes
Ger. J. Vet. Res
2021.
vol. 1, Iss. 4
pp:18-22
Doi: https://doi.org/10.51585/gjvr.2021.4.0026
Abstract:
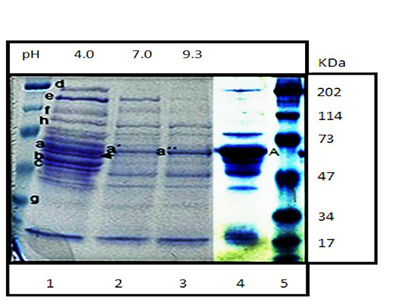
To gain further knowledge of the equine synovial fluid (SF) proteome, we propose a protocol based on the equalization of the relative concentrations of its proteins, which leads to the modification of the standard electrophoretic pattern revealing low-abundance proteins that otherwise remain undetected. Fresh SF samples were collected from ten macroscopically normal metacarpophalangeal joints of crossbred horses. The samples were processed using standard procedures as the control and via combinatorial peptide ligand libraries (CPLL) using low ionic forces (NaH2PO4 10 mM) at different pHs (4.0, 7.0, and 9.3) with 10% sodium dodecyl sulfate (SDS) and 25 mM DTT for protein resolubilization. Proteins were then separated by conventional 8% SDS-PAGE and stained with coomassie blue. After the separation of the equalized proteins, there was a significant reduction in the albumin (the most abundant protein in the SF), and, at the same time, other protein bands arose that were not visible without CPLL processing. In addition, there was variation in the protein profiles at different pHs. The results suggest that protein equalization of the equine SF by CPLL could be a useful tool to better understand articular homeostasis and/or for the detection of new biomarkers of joint pathology
Keywords:
Synovial fluid, Equus caballus, Combinatorial peptide ligand library, Biomarker
Statistics:
Article Views: 2321
PDF Download: 36